
Chemistry, 31.08.2020 02:01 sarrivera579
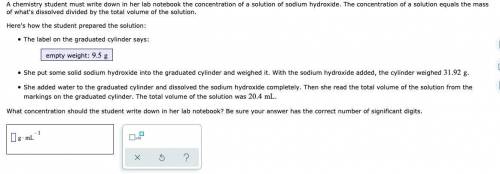
A chemistry student must write down in her lab notebook the concentration of a solution of sodium hydroxide. The concentration of a solution equals the mass of what's dissolved divided by the total volume of the solution. Here's how the student prepared the solution: The label on the graduated cylinder says: empty weight: She put some solid sodium hydroxide into the graduated cylinder and weighed it. With the sodium hydroxide added, the cylinder weighed . She added water to the graduated cylinder and dissolved the sodium hydroxide completely. Then she read the total volume of the solution from the markings on the graduated cylinder. The total volume of the solution was . What concentration should the student write down in her lab notebook? Be sure your answer has the correct number of significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:30
The crust of earth may a- continets and ocean floors. b-continents only. c-layers of sedimentary rocks and continents. d-all of the above
Answers: 2

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

You know the right answer?
A chemistry student must write down in her lab notebook the concentration of a solution of sodium hy...
Questions








Mathematics, 19.06.2020 01:57



Physics, 19.06.2020 01:57

Business, 19.06.2020 01:57

Mathematics, 19.06.2020 01:57



Chemistry, 19.06.2020 01:57







