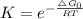Chemistry, 28.08.2020 16:01 j1theking18
Choose the best answer below. Which of the following reactions will have the largest equilibrium constant at 298 K?
a) 302(g) → 203(9) AGOrxn = +326 kJ
b) Mg(s) + N20(g) → Mgo(s) + N2(g) AG9rxn = -673.0 kJ
c) 2Hg(g) + O2(g) → 2HgO(s) AGºrx = -180.8 kJ
d) CaCO3(s) » Cao(s) + CO2(g) AG = +131.1 kJ
It is not possible to determine the reaction with the largest equilibrium constant using the given information.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 11:00
When hydrochloric acid reacts with potassium hydroxide solution, the following reaction occurs. hcl (aq) + koh (aq) h2o (l) + kcl (aq) the reaction gives off heat energy, so it is an reaction.
Answers: 1


Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1

Chemistry, 22.06.2019 20:40
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1
You know the right answer?
Choose the best answer below. Which of the following reactions will have the largest equilibrium con...
Questions

Social Studies, 27.09.2019 22:00



Mathematics, 27.09.2019 22:00

Mathematics, 27.09.2019 22:00

Mathematics, 27.09.2019 22:00






History, 27.09.2019 22:00

Mathematics, 27.09.2019 22:00


English, 27.09.2019 22:00



History, 27.09.2019 22:00


Mathematics, 27.09.2019 22:00