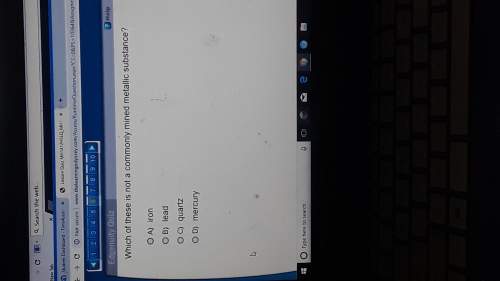
Chemistry, 26.08.2020 01:01 loishsu4936
In Chemistry, a group of students have been presented with 2 test tubes. In both test tubes they observed bubbles rising up in a clear liquid. They are told that hydrogen gas has been produced in one test tube, while carbon dioxide was produced in the other. During this test they have been instructed to collect the gases from each reaction and identify them. How could the students collect and identify the hydrogen gas?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The best solution for preventing harm to people and pets from severe hurricanes involves determining and warning residents about what
Answers: 1

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1


Chemistry, 22.06.2019 21:00
As we move from left to right across the periodic table, what is the general trend? a) atomic radii increase. b) electronegavitiy decreases. c) nuclear shielding increases. d) metallic character decreases.
Answers: 1
You know the right answer?
In Chemistry, a group of students have been presented with 2 test tubes. In both test tubes they obs...
Questions


Mathematics, 08.04.2021 18:10






Mathematics, 08.04.2021 18:10

English, 08.04.2021 18:10


Mathematics, 08.04.2021 18:10


Biology, 08.04.2021 18:10





English, 08.04.2021 18:10