Chemistry, 22.08.2020 01:01 taysomoneyyy
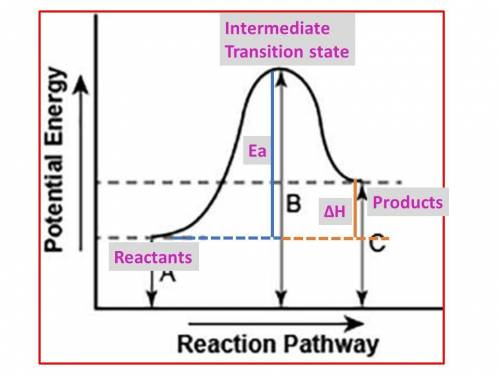
The diagram shows the potential energy changes for a reaction pathway. (10 points) A curve line graph is shown. The y axis of the graph has the title Potential Energy. The x axis of the graph has the title Reaction Pathway. The curve begins at a lower level and ends at a slightly higher level. A vertical line labeled A, starts from the x axis till the beginning of the graph line. A vertical line labeled B starts from the x axis and continues till the peak of the graph. Another vertical line labeled C is shown from the x axis till the point where the curve ends. Part 1: Describe how you can determine the total change in enthalpy and activation energy from the diagram, and if each is positive or negative. Part 2: Describe how the curve will look if the reaction was exothermic. Be sure to mention changes in the potential energies of the reactants and products and the sign changes of the enthalpy.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 17:10
In which block of the periodic table is uranium (u) found? s blockd blockp blockf block
Answers: 1

Chemistry, 23.06.2019 04:00
How much energy is required to vaporize 2 kg of copper? a 4730 kj b 207kj c 9460 kj d 414kj
Answers: 1

Chemistry, 23.06.2019 06:00
In an exothermic reaction at equilibrium, what is the effect of lowering the temperature? a. the reaction makes more products. b. the reaction makes more reactants. c. the reaction is unchanged.
Answers: 1

Chemistry, 23.06.2019 08:00
If the solubility of a gas in water is 1.22 g/l at 2.75 atm, what is its solubility (in g/l) at 1.0 atm?
Answers: 1
You know the right answer?
The diagram shows the potential energy changes for a reaction pathway. (10 points) A curve line grap...
Questions

Mathematics, 05.05.2020 15:09

Mathematics, 05.05.2020 15:09



Physics, 05.05.2020 15:09

English, 05.05.2020 15:09

Social Studies, 05.05.2020 15:09



Mathematics, 05.05.2020 15:09

History, 05.05.2020 15:09

English, 05.05.2020 15:09

Mathematics, 05.05.2020 15:09




Social Studies, 05.05.2020 15:09