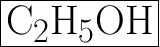Chemistry, 18.08.2020 14:01 bigmouth804
The following compounds have similar molecular weights. Which has the highest boiling point?
A. CH3OCH3
B. C2H5OH
C. CH3CH2CH3
D. CH3CH=O

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Calculate the molar mass of aluminum oxide (al2o3). express your answer to four significant figures.
Answers: 1

Chemistry, 22.06.2019 04:00
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1

Chemistry, 22.06.2019 04:30
Acamcorder has a power rating of 17 watts. if the output voltage from its battery is 7 volts, what current does it use?units:
Answers: 1

Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3
You know the right answer?
The following compounds have similar molecular weights. Which has the highest boiling point?
A. CH3...
Questions

Mathematics, 26.02.2021 14:00

Chemistry, 26.02.2021 14:00


Mathematics, 26.02.2021 14:00


Mathematics, 26.02.2021 14:00

Mathematics, 26.02.2021 14:00

Mathematics, 26.02.2021 14:00

Mathematics, 26.02.2021 14:00

Mathematics, 26.02.2021 14:00

Mathematics, 26.02.2021 14:00

Chemistry, 26.02.2021 14:00



Mathematics, 26.02.2021 14:00

Mathematics, 26.02.2021 14:00

Mathematics, 26.02.2021 14:00

Mathematics, 26.02.2021 14:00

Physics, 26.02.2021 14:00

Mathematics, 26.02.2021 14:00