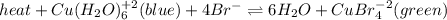The following equilibrium is formed when copper and bromide ions are placed in a solution:
heat + Cu(H2O)6 ^+2 (blue) + 4Br- <--> 6H2O + CuBr4^-2 (green)
A) answer the following questions when KBr is added to the solution:
1. What will happen to the equilibrium?
2. What will be the color of the solution?
3. Will the solution be hotter or cooler? Explain.
B) What will be the color of the solution when the solution is heated?

Answers: 2


Another question on Chemistry

Chemistry, 20.06.2019 18:04
According to the balanced chemical equation 5 h2c2o4(aq) + 2 mno4-(aq) + 6 h+(aq) → 10 co2(g) + 2 mn2+(aq) + 8 h2o(l) 0.875 grams of oxalic acid, h2c2o4 will react with moles of permanganate, mno4-.
Answers: 2

Chemistry, 22.06.2019 08:30
How does the principle of electromagnetism explain the interaction between earth’s magnetic field and the solar wind?
Answers: 1

Chemistry, 22.06.2019 21:30
How many oxygen atoms are there in 3.15 moles of hcl manganese (iv) oxide, mno2
Answers: 2

Chemistry, 23.06.2019 00:30
Arrange the elements in order of increasing electronegativity. use the periodic table to you arrange the elements. p o k mg
Answers: 2
You know the right answer?
The following equilibrium is formed when copper and bromide ions are placed in a solution:
heat + C...
Questions


Mathematics, 12.12.2020 16:10

English, 12.12.2020 16:10


History, 12.12.2020 16:10


Geography, 12.12.2020 16:10


Mathematics, 12.12.2020 16:10


History, 12.12.2020 16:10

English, 12.12.2020 16:10


Business, 12.12.2020 16:10



Mathematics, 12.12.2020 16:10

Mathematics, 12.12.2020 16:10

Mathematics, 12.12.2020 16:10