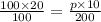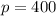Chemistry, 17.08.2020 03:01 orlando19882000
If the temperature of a gas increased from 100K to 200K and the volume of a gas decreased from 20L to 10L, what is the new pressure I’d the original pressure was 100 kPa?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1

Chemistry, 22.06.2019 23:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1


Chemistry, 23.06.2019 03:00
Abaker touches a pie right after taking it out of the oven. which statement best explains why the pie feels hot?
Answers: 2
You know the right answer?
If the temperature of a gas increased from 100K to 200K and the volume of a gas decreased from 20L t...
Questions








Arts, 01.12.2020 01:50

Mathematics, 01.12.2020 01:50



Physics, 01.12.2020 02:00

Mathematics, 01.12.2020 02:00



Social Studies, 01.12.2020 02:00

Biology, 01.12.2020 02:00



Arts, 01.12.2020 02:00