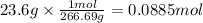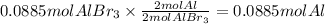Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:30
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible?
Answers: 2

Chemistry, 22.06.2019 07:00
The blackbody curve for a star name zeta is shown below. what is the peak wavelength for this star ?
Answers: 1

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

You know the right answer?
How many moles of Al are necessary to form 23.6 g of AlBr₃ from this reaction: 2 Al(s) + 3 Br₂(l) →...
Questions


Mathematics, 01.10.2019 02:30


Mathematics, 01.10.2019 02:30

History, 01.10.2019 02:30

Physics, 01.10.2019 02:30

Mathematics, 01.10.2019 02:30




English, 01.10.2019 02:30

Biology, 01.10.2019 02:30


English, 01.10.2019 02:30

History, 01.10.2019 02:30



Social Studies, 01.10.2019 02:30