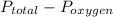Chemistry, 12.08.2020 18:01 Melymarrupe1345
Human lungs have evolved to breathe oxygen at a pressure as that in the atmosphere, 0.21 atm. If a particular heliox mixture to be carried by a scuba diver is at a pressure of 7.00 atm, what should be the partial pressure due to helium in order to maintain the pressure due to oxygen at 0.21 atm?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:00
The agent of mechanical weathering in which rock is worn away by the grinding action of other rock particles is call
Answers: 1

Chemistry, 22.06.2019 12:00
Hey guys so i need to know what is _nh3+> nh4oh ~chemistry~
Answers: 1

Chemistry, 22.06.2019 17:30
Aroller coaster is traveling at 13 mi./s when you purchase a hill that is 400 m long and down the hill exonerate at 4.0 m/s squared what is the final velocity of the posterior found your answer to the nearest number
Answers: 1

Chemistry, 23.06.2019 02:00
Now look at the segment of the graph between the two data points marked with black squares. describe how the boiling point and melting point plots behave between these points. be as specific as possible.
Answers: 1
You know the right answer?
Human lungs have evolved to breathe oxygen at a pressure as that in the atmosphere, 0.21 atm. If a p...
Questions

History, 29.07.2019 01:00









Mathematics, 29.07.2019 01:00

Spanish, 29.07.2019 01:00




Health, 29.07.2019 01:00





Mathematics, 29.07.2019 01:00

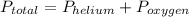 , where
, where 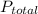 = total partial pressure of all the component gases in the mixture,
= total partial pressure of all the component gases in the mixture, 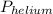 = partial pressure of helium gas, and
= partial pressure of helium gas, and 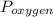 = partial pressure of oxygen gas.
= partial pressure of oxygen gas.