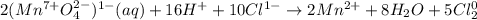Chemistry, 12.08.2020 08:01 Kaziyah461
After balancing the following reaction under acidic conditions, how many mole equivalents of water are required and on which side of the reaction do they appear?
MnO41- (aq) + Cl1- (aq) → Mn2+ (aq) + Cl2 (g)
a. 2 moles of H2O on the reactant side
b. 2 moles of H2O on the product side
c. 4 moles of H2O on the product side
d. 8 moles of H2O on the product side
e. 10 moles of H2O on the reactant side

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:40
Which statement can best be concluded from the ideal gas law?
Answers: 2

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3


Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3
You know the right answer?
After balancing the following reaction under acidic conditions, how many mole equivalents of water a...
Questions

Mathematics, 18.06.2021 14:00

Mathematics, 18.06.2021 14:00





Physics, 18.06.2021 14:00

Biology, 18.06.2021 14:00

Mathematics, 18.06.2021 14:00

English, 18.06.2021 14:00



Biology, 18.06.2021 14:00

Computers and Technology, 18.06.2021 14:00


Biology, 18.06.2021 14:00


English, 18.06.2021 14:00

Physics, 18.06.2021 14:00

Computers and Technology, 18.06.2021 14:00

![MnO_4^{1-} (aq) + Cl^{1-} (aq) \rightarrow Mn^{2+} (aq) + Cl_2 (g)\\\\(Mn^{7+}O^{2-}_4)^{1-} (aq) + Cl^{1-} (aq) \rightarrow Mn^{2+} (aq) + Cl_2 (g)\\\\\\\\(Mn^{7+}O^{2-}_4)^{1-} (aq)+8H^++5e^- \rightarrow Mn^{2+}+4H_2O\\\\2Cl^{1-}\rightarrow Cl_2^0+2e^-\\\\2*[(Mn^{7+}O^{2-}_4)^{1-} (aq)+8H^++5e^- \rightarrow Mn^{2+}+4H_2O]\\\\5*[2Cl^{1-}\rightarrow Cl_2^0+2e^-]\\\\\\\\2(Mn^{7+}O^{2-}_4)^{1-} (aq)+16H^++10e^- \rightarrow 2Mn^{2+}+8H_2O\\\\10Cl^{1-}\rightarrow 5Cl_2^0+10e^-\\](/tpl/images/0720/0206/c85d6.png)