Chemistry, 12.08.2020 07:01 only1cache
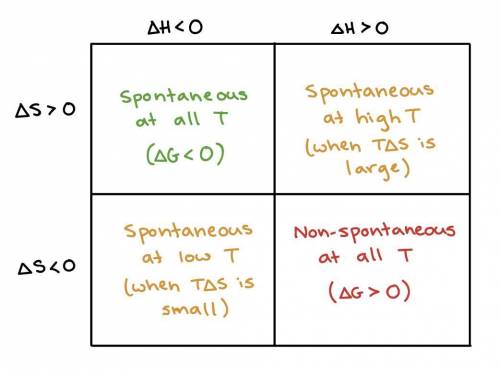
Which of the following combinations will result in a reaction that is spontaneous at all temperatures?
Negative enthalpy change and negative entropy change
Negative enthalpy change and positive entropy change
Positive enthalpy change and negative entropy change
Positive enthalpy change and positive entropy change
PLS EXPLAIN WHAT EACH MEANS AND THE VARIABLES AND THE EXPLANATION BEHIND IT

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 21.06.2019 22:50
Select the correct answer how does the heat content of the reaction change in the process of photosynthesis when a glucose molecule is formed? ca the value of is negative the value of qis positive the value of a remains constant the value of a decreases the value of equals zero e
Answers: 2

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1
You know the right answer?
Which of the following combinations will result in a reaction that is spontaneous at all temperature...
Questions

Mathematics, 19.11.2020 18:20

English, 19.11.2020 18:20

Biology, 19.11.2020 18:20


Advanced Placement (AP), 19.11.2020 18:20

Physics, 19.11.2020 18:20


Mathematics, 19.11.2020 18:20

Mathematics, 19.11.2020 18:20

History, 19.11.2020 18:20










Advanced Placement (AP), 19.11.2020 18:20

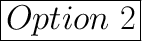
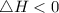 )
) )
)