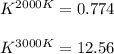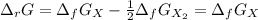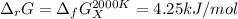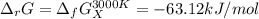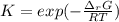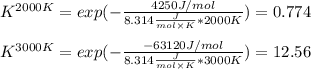Chemistry, 12.08.2020 04:01 anitadefrances
The equation represents the decomposition of a generic diatomic element in its standard state. 12X2(g)⟶X(g) Assume that the standard molar Gibbs energy of formation of X(g) is 4.25 kJ·mol−1 at 2000. K and −63.12 kJ·mol−1 at 3000. K. Determine the value of K (the thermodynamic equilibrium constant) at each temperature.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
14. complete and balance the equations for the single displacement reactions. a. zn + pb(no3)2 -> b. al + niso4 -> 15. complete and balance the equations for the double displacement reactions. a. agno3(aq) + nacl(aq) -> b. mg(no3)2(aq) + koh(aq) -> 16. complete and balance the equations for the combustion reactions. a. __ ch4 + o2 -> b. __ c3h6 + o2 -> c. + o2 ->
Answers: 2

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2

Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1
You know the right answer?
The equation represents the decomposition of a generic diatomic element in its standard state. 12X2(...
Questions


Mathematics, 20.05.2021 16:20

Mathematics, 20.05.2021 16:20

Social Studies, 20.05.2021 16:20

Physics, 20.05.2021 16:20

Mathematics, 20.05.2021 16:20




Chemistry, 20.05.2021 16:20



Mathematics, 20.05.2021 16:20

Biology, 20.05.2021 16:20

Chemistry, 20.05.2021 16:20

Health, 20.05.2021 16:20


Mathematics, 20.05.2021 16:20


Mathematics, 20.05.2021 16:20