
Chemistry, 12.08.2020 06:01 valenzueladomipay09u
Calculate the amount of heat that must be absorbed by 10.0 g of ice at –20°C to convert it to liquid water at 60.0°C. Given: specific heat (ice) = 2.1 J/g·°C; specific heat (water) = 4.18 J/g·°C; ΔH fus = 6.0 kJ/mol.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:40
The formation of a solid, also known as a is an indication of a chemical change. precipitate particulate particle powder
Answers: 3

Chemistry, 22.06.2019 14:00
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1

Chemistry, 22.06.2019 15:30
The gulf stream is a warm water current that flows away from the equator to northern europe. witch of these does it cause. a. crashes of warm and cool water in the ocean b.colder climates near the equator c.large waves on the cost of europe d.warm climates in northern europe
Answers: 1

Chemistry, 22.06.2019 22:30
What is the work done by the electric force to move a 1 c charge from a to b?
Answers: 2
You know the right answer?
Calculate the amount of heat that must be absorbed by 10.0 g of ice at –20°C to convert it to liquid...
Questions




Mathematics, 16.06.2021 20:20

Mathematics, 16.06.2021 20:20

History, 16.06.2021 20:20




Biology, 16.06.2021 20:20

Chemistry, 16.06.2021 20:20



Mathematics, 16.06.2021 20:20


Geography, 16.06.2021 20:20

Mathematics, 16.06.2021 20:20

Mathematics, 16.06.2021 20:20

English, 16.06.2021 20:20

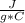 and ΔT=0 C - (-20 C)= 20 C
and ΔT=0 C - (-20 C)= 20 C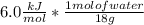 = 3.333 kJ= 3,333 J (being kJ=1,000 J)
= 3.333 kJ= 3,333 J (being kJ=1,000 J)


