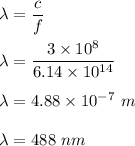Chemistry, 01.08.2020 20:01 waldruphope3956
Given the wavelength of the corresponding emission line, calculate the equivalent radiated energy from n = 4 to n = 2 in both joules and electron volts. Also, calculate the frequency of the wave. λ (Á) = 4861, ƒ(Hz), E(J), E(eV)

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 00:30
Water (2510 g ) is heated until it just begins to boil. if the water absorbs 5.09×105 j of heat in the process, what was the initial temperature of the water?
Answers: 3

Chemistry, 22.06.2019 11:30
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1
You know the right answer?
Given the wavelength of the corresponding emission line, calculate the equivalent radiated energy fr...
Questions


Computers and Technology, 13.07.2019 10:40

Health, 13.07.2019 10:40

Biology, 13.07.2019 10:40


Computers and Technology, 13.07.2019 10:40





Social Studies, 13.07.2019 10:40

Business, 13.07.2019 10:40

Social Studies, 13.07.2019 10:40

Mathematics, 13.07.2019 10:40


Social Studies, 13.07.2019 10:50

Health, 13.07.2019 10:50



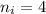
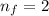
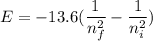
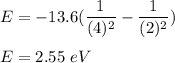
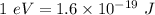
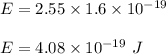
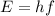
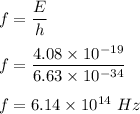
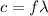
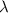 is wavelength
is wavelength