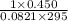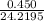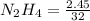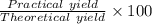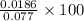Chemistry, 31.07.2020 20:01 gapaxton22
Hydrazine, N2H4 , reacts with oxygen to form nitrogen gas and water. N2H4(aq)+O2(g)⟶N2(g)+2H2O(l) If 2.45 g of N2H4 reacts with excess oxygen and produces 0.450 L of N2 , at 295 K and 1.00 atm, what is the percent yield of the reaction?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
What was the procedure by which case united states vs lopez went to court
Answers: 1

Chemistry, 21.06.2019 21:30
Which is the layer underground where all empty spaces are filled with a combination of air and water ?
Answers: 1

Chemistry, 22.06.2019 10:30
Earth's axis of rotation is tilted at an angle of 23.5 degrees. what is one change you would see on earth if its axis was not tilted?
Answers: 3

Chemistry, 22.06.2019 12:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1
You know the right answer?
Hydrazine, N2H4 , reacts with oxygen to form nitrogen gas and water. N2H4(aq)+O2(g)⟶N2(g)+2H2O(l) If...
Questions


Mathematics, 21.04.2021 01:00

Mathematics, 21.04.2021 01:00

Physics, 21.04.2021 01:00


Mathematics, 21.04.2021 01:00





Mathematics, 21.04.2021 01:00



Mathematics, 21.04.2021 01:00

Mathematics, 21.04.2021 01:00

Geography, 21.04.2021 01:00

Geography, 21.04.2021 01:00


English, 21.04.2021 01:00

Biology, 21.04.2021 01:00

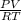
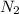 =
=