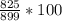Q7. Aluminum is extracted from the ore bauxite, which is impure aluminum oxide. 1 tonne (1000 kg) of the ore was found to have this composition: 1. aluminum oxide 825 kg 2.iron(III) oxide 100 kg 3.sand 75 kg
(a): What percentage of this ore is impurities?
(b): 1 tonne of the ore gives 437 kg of aluminum.
(i): How much aluminum will be obtained from 5 tonnes of the ore?
(ii): What mass of sand is in this 5 tonnes?
(c): What will the percentage of aluminum oxide in the ore be, if all the iron (III) oxide is removed, leaving only the aluminum oxide and sand?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 04:30
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 09:40
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 10:00
The tendency of water molecules to stick together is referred to as a) adhesion b) polarity c) cohesion d) transpiration e) evaporation
Answers: 1
You know the right answer?
Q7. Aluminum is extracted from the ore bauxite, which is impure aluminum oxide. 1 tonne (1000 kg) of...
Questions



Mathematics, 22.07.2019 20:30



Mathematics, 22.07.2019 20:30




Social Studies, 22.07.2019 20:30

English, 22.07.2019 20:30






Mathematics, 22.07.2019 20:30


Mathematics, 22.07.2019 20:30