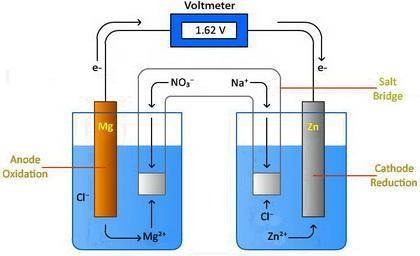
Chemistry, 30.07.2020 08:01 putaprincess16
Design a voltaic cell using magnesium as one of the electrodes. Magnesium can be represented as either Metal A or Metal B in the above drawing. Use metal chlorides as the solutions in the two chambers. For example, magnesium chloride, (MgCl2) will be in solution in the chamber with the magnesium electrode. Use NaNO3 in the salt bridge. Select another element for the other electrode. Explain why you selected this element. Include information about the activity of the metal you select and the need for a spontaneous reaction. Metal A: Metal B: In the drawing, 1. Label the oxidation compartment: 2. Label the reduction compartment. 3. Label the direction of the flow of electrons. 4. Label the flow of the magnesium ions. 5. Label the flow of your selected element's ions. 6. What is leaving the salt bridge in the anode compartment? 7. What is leaving the salt bridge in the cathode compartment? 8. Write the oxidation and reduction half-reactions. 9. Calculate the chemical potential of your cell. Show all of your work.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Needthe meter is the standard unit for: 1) height 2) length 3) weight 4) mass
Answers: 3

Chemistry, 22.06.2019 11:20
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1


Chemistry, 23.06.2019 08:40
Which statement is true according to the kinetic theory? a. molecules of different gases with the same mass and temperature always have the same average density. b. molecules of different gases with the same mass and temperature always have the same average volume. c. molecules of different gases with the same mass and temperature always have the same pressure. d. molecules of different gases with the same mass and temperature always have the same molecular mass. e. molecules of different gases with the same mass and temperature always have the same average kinetic energy.
Answers: 1
You know the right answer?
Design a voltaic cell using magnesium as one of the electrodes. Magnesium can be represented as eith...
Questions

Mathematics, 16.11.2020 02:30










English, 16.11.2020 02:30


Mathematics, 16.11.2020 02:30

English, 16.11.2020 02:30

Mathematics, 16.11.2020 02:30

Mathematics, 16.11.2020 02:30

Mathematics, 16.11.2020 02:30



Biology, 16.11.2020 02:30