
Write a balanced equation for the single-replacement oxidation-reduction reaction described, using the smallest possible integer coefficients. The reaction that takes place when chlorine gas combines with aqueous potassium bromide. (Use the lowest possible coefficients. Omit states of matter.)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:50
An engineering team designs a new rocket that is faster and lighter than any other model being produced. however, the materials end up being so expensive that no company can afford to buy them. which step of the engineering process should have addressed this problem? a. know the background. b. evaluate the results. c. identify a need. d. do the work.
Answers: 2


Chemistry, 22.06.2019 04:30
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1
You know the right answer?
Write a balanced equation for the single-replacement oxidation-reduction reaction described, using t...
Questions


Mathematics, 06.09.2020 03:01

English, 06.09.2020 03:01

Chemistry, 06.09.2020 03:01

Mathematics, 06.09.2020 03:01


History, 06.09.2020 03:01


Mathematics, 06.09.2020 03:01

Mathematics, 06.09.2020 03:01


Medicine, 06.09.2020 03:01

Geography, 06.09.2020 03:01

Arts, 06.09.2020 03:01

Mathematics, 06.09.2020 03:01

History, 06.09.2020 03:01


Spanish, 06.09.2020 03:01


English, 06.09.2020 03:01

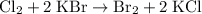 .
.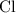 and bromine
and bromine 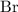 are group 17 elements (halogens.) Each On the other hand, potassium
are group 17 elements (halogens.) Each On the other hand, potassium 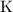 is a group 1 element (alkaline metal.) Each
is a group 1 element (alkaline metal.) Each  . Similarly, the ratio between
. Similarly, the ratio between 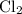 , while the formula for bromine gas is
, while the formula for bromine gas is 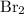 .
.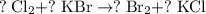 .
.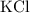 features two elements each.
features two elements each.  .
.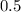 units of
units of 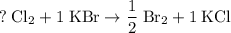 .
.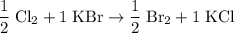 .
.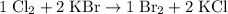 .
.

