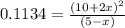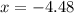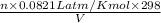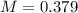Chemistry, 29.07.2020 04:01 leilaneedshelp3395
Consider the equilibrium system: N2O4 (g) = 2 NO2 (g) for which the Kp = 0.1134 at 25 C and deltaH rx is 58.03 kJ/mol. Assume that 1 mole of N2O4 and 2 moles of NO2 are introduced into a 5 L contains. What will be the equilibrium value of [N204]?
A) 0.358 M
B) 0.042 M
C) 0.0822 M
D) 0.928 M
E) 0.379 M

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:30
One mole of zinc has a mass of 65.4 grams. approximately how many atoms of zinc are present in one mole of zinc? 32 × 1023 atoms 6 × 1023 atoms 66 atoms 65 atoms
Answers: 1

Chemistry, 22.06.2019 09:50
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1

Chemistry, 22.06.2019 16:00
How will the volume of a gas be affected if the pressure is tripled, but the temperature remains the same?
Answers: 3

Chemistry, 22.06.2019 21:50
28. which is not a reason that water is used to store spent fuel rods from nuclear power plants? water increases the speed of the chain reaction in the fuel rods. water protects nuclear power plant workers from the high temperature and radiation of the fuel rods. water acts as a radiation shield to reduce the radiation levels. water cools the spent rods. salts action
Answers: 1
You know the right answer?
Consider the equilibrium system: N2O4 (g) = 2 NO2 (g) for which the Kp = 0.1134 at 25 C and deltaH r...
Questions

Mathematics, 30.09.2021 15:50



Mathematics, 30.09.2021 15:50

Mathematics, 30.09.2021 15:50

Social Studies, 30.09.2021 15:50


Medicine, 30.09.2021 15:50

Mathematics, 30.09.2021 15:50

Biology, 30.09.2021 16:00



English, 30.09.2021 16:00


Mathematics, 30.09.2021 16:00

Chemistry, 30.09.2021 16:00




Business, 30.09.2021 16:10

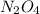 is 0.379 M
is 0.379 M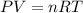
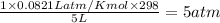
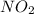 =
= 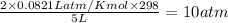
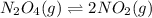
![K_p=\frac{[p_NO_2]^2}{[p_N_2O_4]}](/tpl/images/0714/6163/a9e41.png)