
Chemistry, 29.07.2020 01:01 drubio102004
A multistep reaction can only occur as fast as its slowest step. Therefore, it is the rate law of the slow step that determines the rate law for the overall reaction. Consider the following multistep reaction:
A+B AB(slow)
A+ABA2B(fast
2A+B A2B(overall)
Based on this mechanism, determine the rate law for the overall reaction.
a) rate = kA2BAB
b) rate = kAB
c) rate = kAAB
d) rate = kA2B

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 17:50
You exhale co2 which is produced during cellular respiration. co2 combines with the water in your blood's plasma to make up one half of the body's most important buffer pair, carbonic acid. the more physical activity you engage in, the more co2 your body is producing. you can see this by putting some of the cabbage indicator in a glass and then blowing bubbles into it through a straw. can you see a change in the color of the indicator?
Answers: 2

Chemistry, 22.06.2019 19:40
What type of electromagnetic waves does the human eye see as the colors red blue or green a visible light waves b radio waves c infrared waves d microwaves
Answers: 1

Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2
You know the right answer?
A multistep reaction can only occur as fast as its slowest step. Therefore, it is the rate law of th...
Questions

English, 26.02.2020 23:50

Mathematics, 26.02.2020 23:50

Mathematics, 26.02.2020 23:50



Mathematics, 26.02.2020 23:50

Computers and Technology, 26.02.2020 23:50





Mathematics, 26.02.2020 23:50

History, 26.02.2020 23:50

Computers and Technology, 26.02.2020 23:50


Business, 26.02.2020 23:50




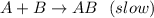
![rate=k[A][B]](/tpl/images/0714/4503/903b2.png)


