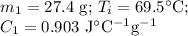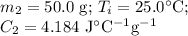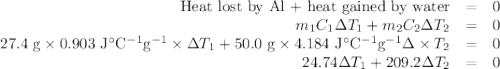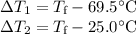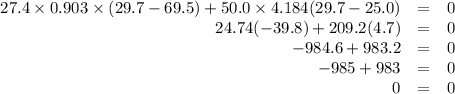Chemistry, 25.07.2020 20:01 kristieroth1
A hot lump of 27.4 g of aluminum at an initial temperature of 69.5 °C is placed in 50.0 mL H2O initially at 25.0 °C and allowed to reach thermal equilibrium. What is the final temperature of the aluminum and water, given that the specific heat of aluminum is 0.903 J/(g·°C)? Assume no heat is lost to surroundings.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2


Chemistry, 22.06.2019 23:00
Need asap question 1 minerals are organic compounds. true false question 2 what vitamin can be found in foods like oranges, grapefruits, and broccoli? a. vitamin a b. vitamin k c.vitamin c d. vitamin d question 3 what are minerals? a. chemical elements that are needed for body processes. b. organic compounds that the body needs in small amounts to function properly. c. small molecules used to build proteins. d. an organic compound that is insoluble in water and includes fats. question 4 how many types of vitamins does the human body need? a. 15 b. 11 c. 13 d. 17 question 5 vitamins are a good source of energy. true false
Answers: 1
You know the right answer?
A hot lump of 27.4 g of aluminum at an initial temperature of 69.5 °C is placed in 50.0 mL H2O initi...
Questions

Mathematics, 05.05.2020 11:10




Mathematics, 05.05.2020 11:10

Mathematics, 05.05.2020 11:10

Social Studies, 05.05.2020 11:10

Chemistry, 05.05.2020 11:10

Mathematics, 05.05.2020 11:10

Mathematics, 05.05.2020 11:10

Social Studies, 05.05.2020 11:10

History, 05.05.2020 11:10



Mathematics, 05.05.2020 11:10

Mathematics, 05.05.2020 11:10

History, 05.05.2020 11:10


Chemistry, 05.05.2020 11:10