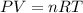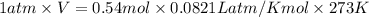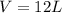Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1

Chemistry, 22.06.2019 13:30
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
How many litres of fluorine gas at stp can be collected from the decomposition of 90.7 g of AuF3...
Questions



Mathematics, 28.02.2021 07:50


Mathematics, 28.02.2021 07:50



Mathematics, 28.02.2021 07:50

Arts, 28.02.2021 07:50


Mathematics, 28.02.2021 07:50


History, 28.02.2021 07:50


Mathematics, 28.02.2021 07:50



Law, 28.02.2021 07:50


Mathematics, 28.02.2021 07:50

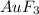
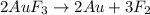
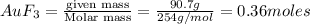
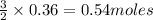 of flourine gas
of flourine gas