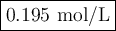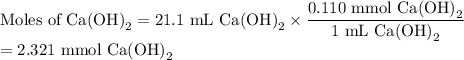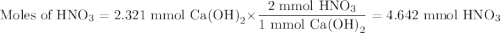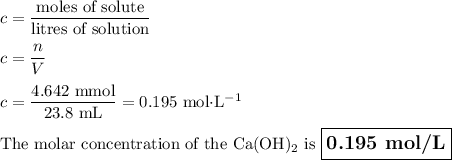Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1
You know the right answer?
An aqueous solution of nitric acid is standardized by titration with a 0.110 M solution of calcium h...
Questions


Mathematics, 28.04.2021 20:10

Mathematics, 28.04.2021 20:10


Mathematics, 28.04.2021 20:10


English, 28.04.2021 20:10


History, 28.04.2021 20:10

English, 28.04.2021 20:10

History, 28.04.2021 20:10

Mathematics, 28.04.2021 20:10

Mathematics, 28.04.2021 20:10

Chemistry, 28.04.2021 20:10

Mathematics, 28.04.2021 20:10


Chemistry, 28.04.2021 20:10