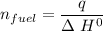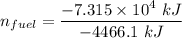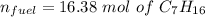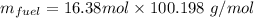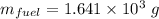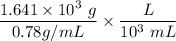Chemistry, 24.07.2020 03:01 daedae11142
When backpacking in the wilderness, hikers often boil water to sterilize it for drinking. Suppose that you are planning a backpacking trip and will need to boil 35 L of water for your group. What volume of fuel should you bring? Assume that the fuel has an average formula of C7 H16 , 15% of the heat generated from combustion goes to heat the water (the rest is lost to the surroundings), the density of the fuel is 0.78 g>mL, the initial temperature of the water is 25.0 °C, and the standard enthalpy of formation of C7 H 16 is -224.4 kJ>mol.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:20
One or more substances changing into one or more substances is an example of a
Answers: 1

Chemistry, 22.06.2019 00:10
Select the correct answer. which phrase correctly describes temperature? o a. average rotational kinetic energy of the particles in an object o b. average energy of the particles in an object c. average translational kinetic energy of the particles in an object od. all energy possessed by the particles in an object
Answers: 1

Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2
You know the right answer?
When backpacking in the wilderness, hikers often boil water to sterilize it for drinking. Suppose th...
Questions






Computers and Technology, 18.03.2021 01:20

Biology, 18.03.2021 01:20

Mathematics, 18.03.2021 01:20




Physics, 18.03.2021 01:20


Mathematics, 18.03.2021 01:20






Social Studies, 18.03.2021 01:20

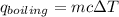
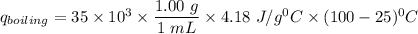
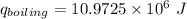
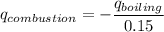
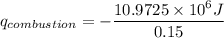
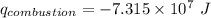
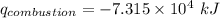
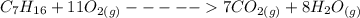
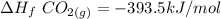
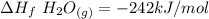
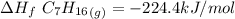
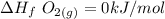
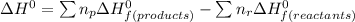
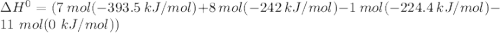
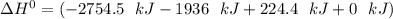
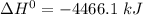
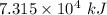 heat released is:
heat released is: