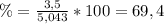1. Unas de las formas de producir nitrógeno gaseoso (N2) es mediante la oxidación de metilamina (CH3NH2), tal como se muestra en la siguiente reacción: CH3NH2 + O2 → CO2 + H2O + N2 Si reaccionan 0,5 mol de metil amina (CH3NH2) con 25,6 g de O2. Determine: a) Balancee la ecuación. (2 ptos) b) ¿Cuántos gramos de nitrógeno (N2) se pueden producir? (4 ptos) c) Si experimentalmente se obtuvieron 3,5 gramos de N2. Determine el porcentaje de rendimiento de la reacción. (4 ptos) Por favor es urgente!!!

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 06:10
56.16 gregor mendel was the first scientist to use statistics to analyze scientific data. before mendel's experiments, scientists believed that organisms acquired traits from their environment and passed them on to their offspring. after mendel's discoveries were accepted, scientists realized that traits passed to offspring were the result of genes being passed from parents to offspring. this is an example of pls
Answers: 1

Chemistry, 22.06.2019 09:30
Right anwser gets marked brainliest newton's discovery concerning how fast an object will change speed is the: 1st law 2nd law 3rd law universal gravitation
Answers: 1

You know the right answer?
1. Unas de las formas de producir nitrógeno gaseoso (N2) es mediante la oxidación de metilamina (CH3...
Questions

History, 18.08.2021 01:00


Engineering, 18.08.2021 01:00


History, 18.08.2021 01:00


Mathematics, 18.08.2021 01:00

Mathematics, 18.08.2021 01:00


Arts, 18.08.2021 01:00

Physics, 18.08.2021 01:00

Advanced Placement (AP), 18.08.2021 01:00


Mathematics, 18.08.2021 01:00

Health, 18.08.2021 01:00

Biology, 18.08.2021 01:00


Mathematics, 18.08.2021 01:00


Mathematics, 18.08.2021 01:00

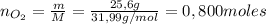
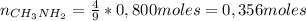
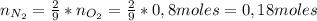
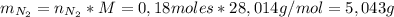
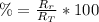
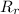 : es el rendimiento real
: es el rendimiento real 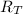 : es el rendimiento teórico
: es el rendimiento teórico