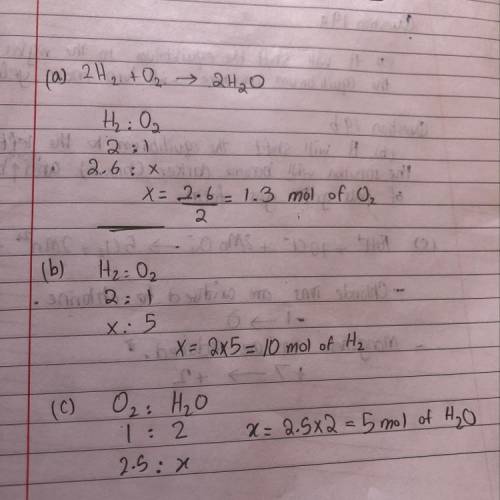The reaction of hydrogen with oxygen produces water.
2H2(g) + O2(8) - 2H2O(g)
a. How many mol...

The reaction of hydrogen with oxygen produces water.
2H2(g) + O2(8) - 2H2O(g)
a. How many moles of O2 are required to react with 2.6 mol
of H2?
b. How many moles of H, are needed to react with 5.0 mol
of O2?
c. How many moles of H2O form when 2.5 mol of O2
reacts?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which one of the following gases is not an important component of soil?
Answers: 2

Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3

Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3

Chemistry, 23.06.2019 01:00
Substance 33°f 100°f peanut oil solid liquid margarine solid liquid chocolate chips solid liquid which conclusion fits the data in the table? a. heat chemically changes chocolate and margarine. b. all solids become liquid at 100°f. c. removing heat from a substance it to melt. d. matter may change shape when it is heated.
Answers: 1
You know the right answer?
Questions

Law, 25.03.2020 18:03


Social Studies, 25.03.2020 18:03

Biology, 25.03.2020 18:03

Geography, 25.03.2020 18:03


Social Studies, 25.03.2020 18:03

Chemistry, 25.03.2020 18:03


Biology, 25.03.2020 18:03

History, 25.03.2020 18:03

Social Studies, 25.03.2020 18:03



Mathematics, 25.03.2020 18:04

History, 25.03.2020 18:13

Mathematics, 25.03.2020 18:13



Mathematics, 25.03.2020 18:13