
1. A 10.0 g sample of propane, C3H8, was combusted in a constant-volume bomb
calorimeter. The total heat capacity of the bomb calorimeter and water was 8.0
kJ/°C. The molar heat of combustion of propane is -2 222 KJ/mol. If the starting
temperature of the water was 20 °C, what will be the final temperature of the
bomb calorimeter?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:00
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2

Chemistry, 22.06.2019 07:20
Describing intermolecular forces use the drop down menus to match the type of intermolecular force to its name dipole dipole interactions dipole induced dipole interactions london dispersion forces hydrogen bond van der waals forces done
Answers: 1


Chemistry, 23.06.2019 04:20
The equation below shows a chemical reaction. a + b + heat —> c + d according to the law of conservation of energy, which statement is true? a. the reactants absorb heat because they have less energy than the products. b. the products release heat because they have more energy than the reactants. c. the reactants generate heat because they have more energy than the products. d. the products require heat to form because they have less energy than the reactants.
Answers: 1
You know the right answer?
1. A 10.0 g sample of propane, C3H8, was combusted in a constant-volume bomb
calorimeter. The total...
Questions


Mathematics, 30.09.2020 04:01

Mathematics, 30.09.2020 04:01

English, 30.09.2020 04:01


Biology, 30.09.2020 04:01


Mathematics, 30.09.2020 04:01




Mathematics, 30.09.2020 04:01

History, 30.09.2020 04:01

English, 30.09.2020 04:01

Mathematics, 30.09.2020 04:01

Social Studies, 30.09.2020 04:01


Mathematics, 30.09.2020 04:01

Geography, 30.09.2020 04:01

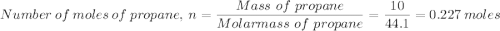 Which gives;
Which gives; in the calorimeter = Heat capacity × Temperature change
in the calorimeter = Heat capacity × Temperature change


