Chemistry, 20.07.2020 01:01 cheesecake1919
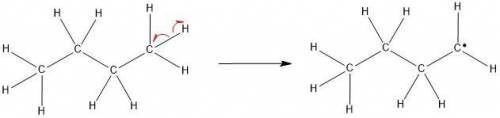
The molecular formula of butane is C4H10. It is obtained from petroleum and is used commonly in LPG (Liquefied Petroleum Gas) cylinders (a common source of cooking gas). It has two arrangements of carbon atoms: a straight chain and a branched chain. Using this information, draw the structure of the tertiary butyl radical that will form upon removal of a hydrogen atom.
Requried:
Draw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars. Include all free radicals by right-clicking on an atom on the canvas and then using the Atom properties to select the monovalent radical.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Often on a topographic map, every fifth contour line is darkened. what is this line called? a. key b.slope c.benchmark d. index contour
Answers: 1

Chemistry, 22.06.2019 01:30
An empty fuel tank can still contain and therefore can be even more dangerous than one full of liquid fuel.
Answers: 1

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

You know the right answer?
The molecular formula of butane is C4H10. It is obtained from petroleum and is used commonly in LPG...
Questions


Mathematics, 23.03.2021 14:00


Mathematics, 23.03.2021 14:00

English, 23.03.2021 14:00

Health, 23.03.2021 14:00

Mathematics, 23.03.2021 14:00



Mathematics, 23.03.2021 14:00

English, 23.03.2021 14:00

Health, 23.03.2021 14:00

English, 23.03.2021 14:00

Health, 23.03.2021 14:00






Mathematics, 23.03.2021 14:00