
Chemistry, 19.07.2020 01:01 josephfoxworth
A solution contains 0.0440 M Ca2 and 0.0940 M Ag. If solid Na3PO4 is added to this mixture, which of the phosphate species would precipitate out of solution first?
A. Na3PO4.
B. Ag3PO4.
C. Ca3(PO4)2
When the second cation just starts to precipitate, what percentage of the first cation remains in solution?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 09:30
What are scientists who study fossils called? ( a ) astronomers. ( b ) biologists. ( c ) geologists. ( d ) paleontologists.
Answers: 2

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 22.06.2019 20:00
How are the terms group and period used on the periodic table
Answers: 1
You know the right answer?
A solution contains 0.0440 M Ca2 and 0.0940 M Ag. If solid Na3PO4 is added to this mixture, which of...
Questions

Mathematics, 06.05.2020 04:33


Mathematics, 06.05.2020 04:33



Social Studies, 06.05.2020 04:33



Mathematics, 06.05.2020 04:33










Biology, 06.05.2020 04:33

English, 06.05.2020 04:33

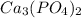 will precipitate out first
will precipitate out first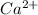 remaining = 12.86%
remaining = 12.86%![[Ca^{2+}] = 0.0440 \ M](/tpl/images/0709/5794/07981.png)
![[Ag^+] = 0.0940 \ M](/tpl/images/0709/5794/e77de.png)
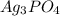
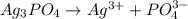
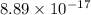
![Ksp = [Ag^+]^3[PO_4^{3-}]](/tpl/images/0709/5794/f170d.png)
![8.89 \times 10 ^{-17} = (0.0940)^3[PO_4^{3-}]](/tpl/images/0709/5794/f1b1a.png)
![\dfrac{8.89 \times 10 ^{-17}}{(0.0940)^3} = [PO_4^{3-}]](/tpl/images/0709/5794/fb0ea.png)
![[PO_4^{3-}] =\dfrac{8.89 \times 10 ^{-17}}{(0.0940)^3}](/tpl/images/0709/5794/36b7f.png)
![[PO_4^{3-}] =1.07 \times 10^{-13}](/tpl/images/0709/5794/d9e0c.png)
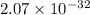
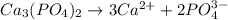
![Ksp = [Ca^{2+}]^3 [PO_4^{3-}]^2](/tpl/images/0709/5794/409cc.png)
![2.07 \times 10^{-33} = (0.0440)^3 [PO_4^{3-}]^2](/tpl/images/0709/5794/a627b.png)
![\dfrac{2.07 \times 10^{-33} }{(0.0440)^3}= [PO_4^{3-}]^2](/tpl/images/0709/5794/4ad8e.png)
![[PO_4^{3-}]^2 = \dfrac{2.07 \times 10^{-33} }{(0.0440)^3}](/tpl/images/0709/5794/fbe62.png)
![[PO_4^{3-}]^2 = 2.43 \times 10^{-29}](/tpl/images/0709/5794/43b90.png)
![[PO_4^{3-}] = \sqrt{2.43 \times 10^{-29}](/tpl/images/0709/5794/0afa0.png)
![[PO_4^{3-}] =4.93 \times 10^{-15}](/tpl/images/0709/5794/19717.png)
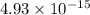 in
in 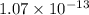
![[Ca^+]](/tpl/images/0709/5794/2b2af.png) when the second cation starts to precipitate ; we have :
when the second cation starts to precipitate ; we have :![2.07 \times 10^{-33} = [Ca^{2+}]^3 (1.07 \times 10^{-13})^2](/tpl/images/0709/5794/8e719.png)
![[Ca^{2+}]^3 = \dfrac{2.07 \times 10^{-33} }{(1.07 \times 10^{-13})^2}](/tpl/images/0709/5794/5de75.png)
![[Ca^{2+}]^3 =1.808 \times 10^{-7}](/tpl/images/0709/5794/9d3e5.png)
![[Ca^{2+}] =\sqrt[3]{1.808 \times 10^{-7}}](/tpl/images/0709/5794/21597.png)
![[Ca^{2+}] =0.00566](/tpl/images/0709/5794/67420.png)
![[Ca^{2+}]](/tpl/images/0709/5794/17576.png) in the solution is 0.00566
in the solution is 0.00566

