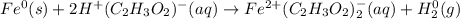Chemistry, 16.07.2020 04:01 danielwu462
Fe(s)+ 2 HC2H3O2(aq) → Fe(C2H3O2)2(aq) + H2(g) Which substance is reduced and which subject is oxidized?

Answers: 1


Another question on Chemistry

Chemistry, 20.06.2019 18:02
Chemistry answer as much as you can if you can't answer more then 2 don't answer
Answers: 2

Chemistry, 22.06.2019 00:40
Base your answer on the information below and on your knowledge of chemistry. nitrogen dioxide, no2, is a dark brown gas that is used to make nitric acid and to bleach flour. nitrogen dioxide has a boiling point of 294 k at 101.3 kpa. in a rigid cylinder with a movable piston, nitrogen dioxide can be in equilibrium with colorless dinitrogen tetroxide, n2o4. this equilibrium is represented by the equation below. 2no2(g) n2o4(g) + 58kj at standard pressure, compare the strength of intermolecular forces in no2(g) to the strength of intermolecular forces in n2(g).
Answers: 2

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 09:00
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2
You know the right answer?
Fe(s)+ 2 HC2H3O2(aq) → Fe(C2H3O2)2(aq) + H2(g) Which substance is reduced and which subject is oxidi...
Questions


Mathematics, 28.01.2021 02:20


History, 28.01.2021 02:20




English, 28.01.2021 02:20

Biology, 28.01.2021 02:20


Mathematics, 28.01.2021 02:20




Mathematics, 28.01.2021 02:20