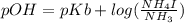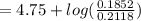Chemistry, 15.07.2020 02:01 kevinvalencia01
An aqueous solution contains 0.397 M ammonia. Calculate the pH of the solution after the addition of 4.63 x 10-2 moles of perchloric acid (HClO4) to 250 mL of this solution. (Assume the volume does not change upon adding perchloric acid). Ka = 5.7 x 10-10, Kb = 1.80 x 10-5

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 13:00
The number of neutrons is equal to the atomic number minus the atomic mass. a. true b. false
Answers: 2

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
You know the right answer?
An aqueous solution contains 0.397 M ammonia. Calculate the pH of the solution after the addition of...
Questions


Mathematics, 29.03.2021 22:10



English, 29.03.2021 22:10


Chemistry, 29.03.2021 22:10


English, 29.03.2021 22:10


Mathematics, 29.03.2021 22:10

Mathematics, 29.03.2021 22:10




Mathematics, 29.03.2021 22:10

Spanish, 29.03.2021 22:10

Mathematics, 29.03.2021 22:10

Mathematics, 29.03.2021 22:10

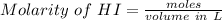
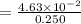
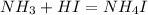
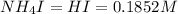
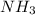 left is
left is