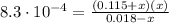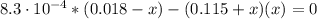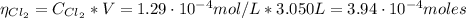Chemistry, 14.07.2020 23:01 4801651052
: Starting with 0.3500 mol CO(g) and 0.05500 mol COCl2(g) in a 3.050-L flask at 668 K, how many moles of Cl2(g) will be present at equilibrium

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1

Chemistry, 23.06.2019 00:30
Balance the following reaction. as2s3 + 9o2 → 2as2o3 + so2
Answers: 2

Chemistry, 23.06.2019 06:30
Acertain atom has 22 protons and 19 electrons. this atom loses an electron. the net charge on the atom is now 4+1+01-4-. if this same atom with 22 protons and 19 electrons were to gain 3 electrons, the net charge on the atom would be 3+2+02-3-.
Answers: 1
You know the right answer?
: Starting with 0.3500 mol CO(g) and 0.05500 mol COCl2(g) in a 3.050-L flask at 668 K, how many mole...
Questions


English, 29.11.2021 03:30


Mathematics, 29.11.2021 03:30

Mathematics, 29.11.2021 03:30





Mathematics, 29.11.2021 03:30

Mathematics, 29.11.2021 03:30

Mathematics, 29.11.2021 03:30

Physics, 29.11.2021 03:30

Mathematics, 29.11.2021 03:30


Mathematics, 29.11.2021 03:30




Mathematics, 29.11.2021 03:30

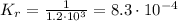
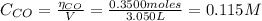
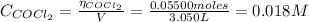
![K_{r} = \frac{[CO][Cl_{2}]}{[COCl_{2}]}](/tpl/images/0706/1416/cc018.png)