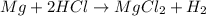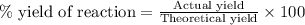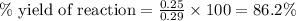Chemistry, 14.07.2020 22:01 dixonmckenzie4636
A student measures a pressure increase of 0.25 atm while collecting the H2 gas products from the reaction of Mg with HCl. The theoretical pressure change is 0.29 atm. What is the % yield

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Your friend offers to show you an intrusive igneous rock. which of the following would you expect to see?
Answers: 1

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1


Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3
You know the right answer?
A student measures a pressure increase of 0.25 atm while collecting the H2 gas products from the rea...
Questions




Mathematics, 23.01.2020 14:31


Social Studies, 23.01.2020 14:31

Biology, 23.01.2020 14:31


Mathematics, 23.01.2020 14:31

Mathematics, 23.01.2020 14:31

Social Studies, 23.01.2020 14:31


Mathematics, 23.01.2020 14:31


English, 23.01.2020 14:31

History, 23.01.2020 14:31

Mathematics, 23.01.2020 14:31

Geography, 23.01.2020 14:31


Social Studies, 23.01.2020 14:31