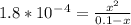Chemistry, 07.07.2020 19:01 rosie20052019
Calculate the pH for the following 1.0M weak acid solutions:a. HCOOH Ka = 1.8 x 10-4 [

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 23.06.2019 00:00
The graph indicates the running route for tobias. which best describes his run? from time 0 to 6, he went fast and then slowed down. from time 6 to 10, he was at his slowest. from time 12 to 14, he went very slow. from time 14 to 18, he went toward the starting point.
Answers: 2
You know the right answer?
Calculate the pH for the following 1.0M weak acid solutions:a. HCOOH Ka = 1.8 x 10-4 [...
Questions




History, 29.06.2019 19:30


Spanish, 29.06.2019 19:30


Mathematics, 29.06.2019 19:30




Mathematics, 29.06.2019 19:30



Computers and Technology, 29.06.2019 19:30

History, 29.06.2019 19:30

English, 29.06.2019 19:30



History, 29.06.2019 19:30

![K_{a} =\frac{product}{reactant}=\frac{[H^+][HCOO^-]}{[HCOOH]}](/tpl/images/0702/5573/bdea7.png) .
.