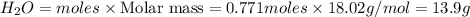Chemistry, 05.07.2020 23:01 stefancvorovic1
How many grams of H 2O are produced from 28.8 g of O 2? (Molar Mass of H 2O = 18.02 g) (Molar Mass of O 2=32.00 g) 4 NH 3 (g) + 7 O 2 (g) → 4 NO 2 (g) + 6 H 2O (g)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:50
An engineering team designs a new rocket that is faster and lighter than any other model being produced. however, the materials end up being so expensive that no company can afford to buy them. which step of the engineering process should have addressed this problem? a. know the background. b. evaluate the results. c. identify a need. d. do the work.
Answers: 2


Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1

Chemistry, 22.06.2019 19:10
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1
You know the right answer?
How many grams of H 2O are produced from 28.8 g of O 2? (Molar Mass of H 2O = 18.02 g) (Molar Mass o...
Questions

Mathematics, 09.04.2021 02:10

English, 09.04.2021 02:10

Mathematics, 09.04.2021 02:10

Health, 09.04.2021 02:10


Biology, 09.04.2021 02:10


English, 09.04.2021 02:10

Mathematics, 09.04.2021 02:10


Mathematics, 09.04.2021 02:10







Mathematics, 09.04.2021 02:10

Computers and Technology, 09.04.2021 02:10

Biology, 09.04.2021 02:10

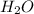 will be produced from the given mass of oxygen
will be produced from the given mass of oxygen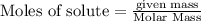
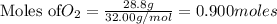
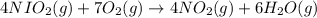
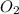 produce = 6 moles of
produce = 6 moles of 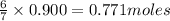 of
of