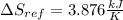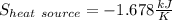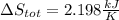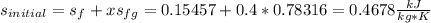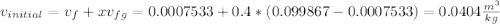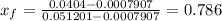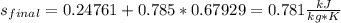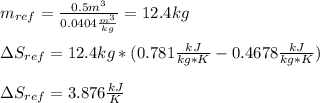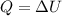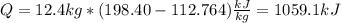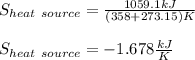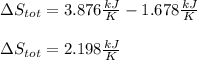A 0.5-m3 rigid tank contains refrigerant-134a initially at 200 kPa and 40 percent quality. Heat is transferred now to the refrigerant from a source at 358C until the pressure rises to 400 kPa. Determine (a) the entropy change of the refrigerant, (b) the entropy change of the heat source, and (c) the total entropy change for this process.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no2]2 ? second order 3/2 order third order zero order none of the listed answers are correct
Answers: 3

Chemistry, 22.06.2019 03:00
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3

Chemistry, 22.06.2019 10:00
The tendency of water molecules to stick together is referred to as a) adhesion b) polarity c) cohesion d) transpiration e) evaporation
Answers: 1
You know the right answer?
A 0.5-m3 rigid tank contains refrigerant-134a initially at 200 kPa and 40 percent quality. Heat is t...
Questions

Computers and Technology, 24.07.2019 01:30

History, 24.07.2019 01:30

History, 24.07.2019 01:30

Physics, 24.07.2019 01:30

Advanced Placement (AP), 24.07.2019 01:30


Biology, 24.07.2019 01:30


History, 24.07.2019 01:30

Biology, 24.07.2019 01:30

History, 24.07.2019 01:30

History, 24.07.2019 01:30

Health, 24.07.2019 01:30

Social Studies, 24.07.2019 01:30

Health, 24.07.2019 01:30

Health, 24.07.2019 01:30

Health, 24.07.2019 01:30

Health, 24.07.2019 01:30

Health, 24.07.2019 01:30

Health, 24.07.2019 01:30