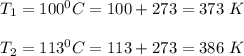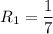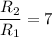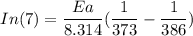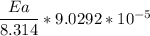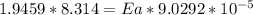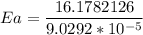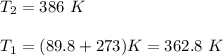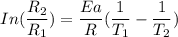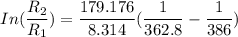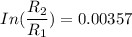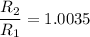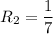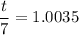Chemistry, 04.07.2020 14:01 landonp101
The water in a pressure cooker boils at a temperature greater than 100°C because it is under pressure. At this higher temperature, the chemical reactions associated with the cooking of food take place at a greater rate. (a) Some food cooks fully in 7.00 min in a pressure cooker at 113.0°C and in 49.0 minutes in an open pot at 100.0°C. Calculate the average activation energy for the reactions associated with the cooking of this food. kJ mol-1 (b) How long will the same food take to cook in an open pot of boiling water at an altitude of 10000 feet, where the boiling point of water is 89.8 °C? min

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
What is the percentage composition of carbon in the compound ch4
Answers: 1

Chemistry, 22.06.2019 11:00
As air becomes more dense, (select all that apply) o. air weighs less o. gas molecules are closer together o. air is colder o. air weighs more o. gas molecules are further apart o. air is hotter
Answers: 3


Chemistry, 22.06.2019 11:30
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3
You know the right answer?
The water in a pressure cooker boils at a temperature greater than 100°C because it is under pressur...
Questions

Mathematics, 02.08.2019 21:00

History, 02.08.2019 21:00

English, 02.08.2019 21:00




Mathematics, 02.08.2019 21:00





Mathematics, 02.08.2019 21:00




Mathematics, 02.08.2019 21:00

Chemistry, 02.08.2019 21:00

Biology, 02.08.2019 21:00

Mathematics, 02.08.2019 21:00