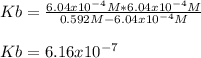Chemistry, 03.07.2020 23:01 mariposa91
In the laboratory, a general chemistry student measured the pH of a 0.592 M aqueous solution of triethanolamine, C6H15O3N to be 10.781. Use the information she obtained to determine the Kb for this base. Kb(experiment) =

Answers: 3


Another question on Chemistry

Chemistry, 23.06.2019 00:30
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2

Chemistry, 23.06.2019 01:30
Magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. trial 1: trial 2: data mass of empty crucible with lid trial 1: 26.688 trial 2: 26.681 mass of mg metal, crucible, and lid trial 1: 26.994 trial: 2 26.985 mass of mgo, crucible, and lid trial 1: 27.188 trial 2: 27.180
Answers: 1

Chemistry, 23.06.2019 02:00
An alpha particle is: a hydrogen atom a nucleus of helium two neutrons an electron
Answers: 1

Chemistry, 23.06.2019 07:00
What are the trends and exceptions to the trends in electron affinity?
Answers: 1
You know the right answer?
In the laboratory, a general chemistry student measured the pH of a 0.592 M aqueous solution of trie...
Questions

Mathematics, 24.08.2021 22:00

Mathematics, 24.08.2021 22:00

Mathematics, 24.08.2021 22:00


Mathematics, 24.08.2021 22:00

Chemistry, 24.08.2021 22:00


French, 24.08.2021 22:00


Spanish, 24.08.2021 22:00

English, 24.08.2021 22:00

Biology, 24.08.2021 22:00


Social Studies, 24.08.2021 22:00

Computers and Technology, 24.08.2021 22:00

English, 24.08.2021 22:00


English, 24.08.2021 22:00

History, 24.08.2021 22:00

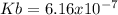
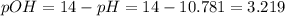
![pOH=-log([OH^-])](/tpl/images/0700/9022/4105a.png)
![[OH^-]=10^{-pOH}=10^{-3.219}=6.04x10^{-4}M](/tpl/images/0700/9022/a7a1d.png)
![Kb=\frac{[OH^-][C6H14O2N^+]}{[C6H15O3N ]}](/tpl/images/0700/9022/e215d.png)
 equals the concentration of hydroxyl ions that was previously, computed, therefore, we have:
equals the concentration of hydroxyl ions that was previously, computed, therefore, we have: