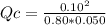The reaction of nitrogen gas and oxygen gas to form nitrogen monoxide gas is shown below. If the measured concentrations of all three chemicals at some point in time are: [N2] = 0.80 M
[O2] = 0.050 M
[NO] = 0.10 M
Which statement is TRUE about the reaction at this point in time? N2(g) + O2(g) ⇄ 2 NO(g) K = 0.10
The reaction is at equilibrium.
The reverse reaction is occurring at a faster rate than the forward reaction.
The forward reaction is occurring at a faster rate than the reverse reaction.
This set of concentration values is impossible because the concentrations of N2 and O2 must be the same.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 22.06.2019 05:30
What happens to the atomic radius when an elctron is lost
Answers: 1


Chemistry, 22.06.2019 12:00
Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution of sodium nitrate. a solution containing 3.50 g of sodium carbonate is mixed with one containing 5.00 g of silver nitrate. how many grams of sodium carbonate, silver nitrate, silver carbonate, and sodium nitrate are present after the reaction is complete?
Answers: 2
You know the right answer?
The reaction of nitrogen gas and oxygen gas to form nitrogen monoxide gas is shown below. If the mea...
Questions


Mathematics, 12.10.2021 01:50

Mathematics, 12.10.2021 01:50

Computers and Technology, 12.10.2021 01:50

Mathematics, 12.10.2021 01:50

Mathematics, 12.10.2021 01:50

Business, 12.10.2021 01:50

English, 12.10.2021 01:50

English, 12.10.2021 01:50


Mathematics, 12.10.2021 01:50

Spanish, 12.10.2021 01:50


Social Studies, 12.10.2021 01:50

Mathematics, 12.10.2021 01:50

English, 12.10.2021 01:50

English, 12.10.2021 01:50



![Qc=\frac{[C]^{c}*[D]^{d} }{[A]^{a}*[B]^{b} }](/tpl/images/0700/8667/038b9.png)
![Qc=\frac{[NO]^{2} }{[N_{2} ]*[O_{2} ] }](/tpl/images/0700/8667/47464.png)