Chemistry, 28.06.2020 02:01 rachiegonzo7
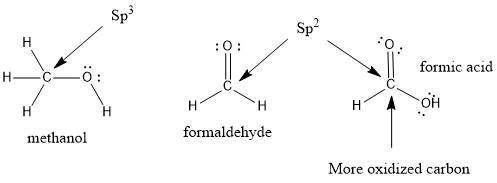
Draw the Lewis structures for CH3OH, CH2O and HCOOH. Indicate the hybrid orbital used in the sigma bonds for each of the carbon atoms. For which of these compounds is the extent of oxidation the greatest? Explain why

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2


Chemistry, 22.06.2019 05:30
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 10:10
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1
You know the right answer?
Draw the Lewis structures for CH3OH, CH2O and HCOOH. Indicate the hybrid orbital used in the sigma b...
Questions

Mathematics, 06.11.2020 01:00

Mathematics, 06.11.2020 01:00


Health, 06.11.2020 01:00


Health, 06.11.2020 01:00

Mathematics, 06.11.2020 01:00

Mathematics, 06.11.2020 01:00

Mathematics, 06.11.2020 01:00



Mathematics, 06.11.2020 01:00


History, 06.11.2020 01:00

Mathematics, 06.11.2020 01:00

Chemistry, 06.11.2020 01:00



English, 06.11.2020 01:00

SAT, 06.11.2020 01:00

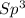 => 4 single bonds
=> 4 single bonds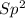 => 1 double bond
=> 1 double bond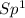 => 1 double bond
=> 1 double bond