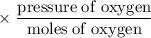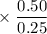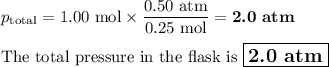Chemistry, 25.06.2020 06:01 kamillecronk
Question 8
1 pts
A closed flask contains a 0.25 moles of O2 which exerts a pressure of
0.50 atm. If 0.75 moles of CO, is added to the container what is the
total pressure in the flask?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 08:30
What are the first three quantum numbers for the electrons located in subshell 2s?
Answers: 2

Chemistry, 22.06.2019 14:30
Which of the following represents the ester functional group? a. -coo- b. -cho c. -cooh d. c=o
Answers: 1

Chemistry, 22.06.2019 18:00
Which statement best describes the he properties of iconic compounds ?
Answers: 1
You know the right answer?
Question 8
1 pts
A closed flask contains a 0.25 moles of O2 which exerts a pressure of
0....
A closed flask contains a 0.25 moles of O2 which exerts a pressure of
0....
Questions


Mathematics, 17.10.2020 04:01

English, 17.10.2020 04:01

Medicine, 17.10.2020 04:01

Mathematics, 17.10.2020 04:01


Mathematics, 17.10.2020 04:01

Chemistry, 17.10.2020 04:01

Mathematics, 17.10.2020 04:01




Mathematics, 17.10.2020 04:01

Mathematics, 17.10.2020 04:01

Mathematics, 17.10.2020 04:01

Mathematics, 17.10.2020 04:01


Mathematics, 17.10.2020 04:01


Medicine, 17.10.2020 04:01