An aqueous solution contains
32.7% KCI (weight/weight %).
What is the mole fraction of KCI in...

Chemistry, 25.06.2020 02:01 angelequej1167
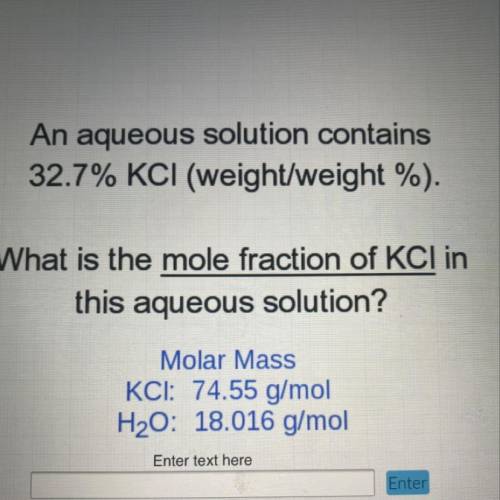
An aqueous solution contains
32.7% KCI (weight/weight %).
What is the mole fraction of KCI in
this aqueous solution?
Molar Mass
KCI: 74.55 g/mol
H2O: 18.016 g/mol
Enter text here
Enter

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:00
What postulate of the kinetic molecular theory best explains why gases have high fluidity? because collisions between gas particles are elastic, there is no loss of energy as particles flow past each other. because gases consist of large numbers of tiny particles, they spread out and do not come in contact with each other. because the attractive forces between gas particles are negligible, gas particles can glide easily past one another. because the average kinetic energy of gas particles increases as temperature increases, gas particles behave more like a liquid. question 6 compare the compressibility of gases and liquids. support your answer by describing the arrangement of particles in gases and liquids.
Answers: 1

Chemistry, 22.06.2019 00:00
Which of the following statements is true? a. elements in the last period are radioactive. b. atomic weight is the same as atomic mass. c. elements in the same group have the same number of electron shells. d. atomic number equals the number of neutrons in the nucleus of an atom.
Answers: 1


Chemistry, 22.06.2019 22:30
Akno3 solution containing 51 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 3
You know the right answer?
Questions

Geography, 22.09.2019 11:00

History, 22.09.2019 11:00

History, 22.09.2019 11:00

Mathematics, 22.09.2019 11:00

Mathematics, 22.09.2019 11:00


Mathematics, 22.09.2019 11:00

Spanish, 22.09.2019 11:00

Mathematics, 22.09.2019 11:00

English, 22.09.2019 11:00




Mathematics, 22.09.2019 11:00


History, 22.09.2019 11:00


Health, 22.09.2019 11:00




