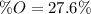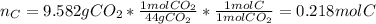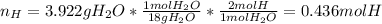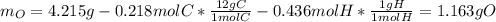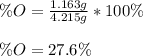Chemistry, 20.06.2020 17:57 collin0123
A 4.215 g sample of a compound containing only carbon, hydrogen, and oxygen is burned in an excess of oxygen gas, producing 9.582 g CO2 and 3.922 g H2O. What percent by mass of oxygen is contained in the original sample?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 22:30
Essay-alternative energy sources research sources of energy that are being developed. write a report of 350-400 words discussing the information you learned concerning the development of various energy sources and the impact that you think they will have on your life. include sources cited at the end of your report using the mla format. follow the rubric guidelines. note that wikipedia is not an appropriate resource for a research paper. worth 99
Answers: 3


Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1

Chemistry, 23.06.2019 01:30
Which statement justifies that hydrogen peroxide (h2o2) is a polar molecule? the o – h bond is nonpolar and the molecule is asymmetric. the o – h bond is nonpolar and the molecule is symmetric. the o – h bond is polar and the molecule is asymmetric. the o – h bond is polar and the molecule is symmetric.
Answers: 1
You know the right answer?
A 4.215 g sample of a compound containing only carbon, hydrogen, and oxygen is burned in an excess o...
Questions

English, 06.12.2021 22:40

Computers and Technology, 06.12.2021 22:40


Mathematics, 06.12.2021 22:40




Biology, 06.12.2021 22:40


Computers and Technology, 06.12.2021 22:40


English, 06.12.2021 22:40


Chemistry, 06.12.2021 22:40




Mathematics, 06.12.2021 22:40

Mathematics, 06.12.2021 22:40

English, 06.12.2021 22:40