
Chemistry, 20.06.2020 08:57 graciewyatt471
1) How many kJ are absorbed when 45.2 g of water at 31.3 oC is heated to 76.9 oC? 2) Calculate the total heat released in kcal when 72.1 g water at 25.2 oC is cooled to 0 oC and freezes. 3) How many kilojoules are required to heat 55,500 mg of gold with specific heat = 0.129 J/g oC is heated from 24.6 oC to 123.4 oC? 4) Calculate the heat needed in kcal to change 45.6 g of water at 100 oC to change into steam.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2
You know the right answer?
1) How many kJ are absorbed when 45.2 g of water at 31.3 oC is heated to 76.9 oC? 2) Calculate the t...
Questions

Social Studies, 26.10.2020 07:20


Mathematics, 26.10.2020 07:20

Mathematics, 26.10.2020 07:20

History, 26.10.2020 07:20

Social Studies, 26.10.2020 07:20


History, 26.10.2020 07:20

Mathematics, 26.10.2020 07:20

Arts, 26.10.2020 07:20

History, 26.10.2020 07:20

Advanced Placement (AP), 26.10.2020 07:20


Mathematics, 26.10.2020 07:20


Mathematics, 26.10.2020 07:20


Mathematics, 26.10.2020 07:20


English, 26.10.2020 07:20

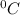 and Δθ is the change in temperature.
and Δθ is the change in temperature.

