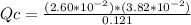Chemistry, 19.06.2020 03:57 ericchen4399
Consider the following reaction where Kc = 1.20×10-2 at 500 K. PCl5(g) PCl3(g) + Cl2(g) A reaction mixture was found to contain 0.121 moles of PCl5(g), 2.60×10-2 moles of PCl3(g), and 3.82×10-2 moles of Cl2(g), in a 1.00 liter container. Is the reaction at equilibrium? If not, what direction must it run in order to reach equilibrium? The reaction quotient, Qc, equals . The reaction A. must run in the forward direction to reach equilibrium. B. must run in the reverse direction to reach equilibrium. C. is at equilibrium.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1

Chemistry, 22.06.2019 16:00
No copying 15 pts how does a free-body diagram tell you about the net force on an object?
Answers: 2

Chemistry, 22.06.2019 18:30
Two people each hold the end of a rope and create waves by moving their arms up and down. this wave is best classified as a transverse wave because a) both the rope particles and the wave are moving in the same direction. b) the wave is moving up and down as the particles of the rope move horizontally. c) the wave is moving horizontally as the particles of the rope move up and down. eliminate d) the wave is moving in a parallel direction with the motion of the person's arms.
Answers: 3

Chemistry, 23.06.2019 04:31
One student said that the investigation was not valid (a fair test). write a plan for the investigation that includes improvements to the method and apparatus
Answers: 1
You know the right answer?
Consider the following reaction where Kc = 1.20×10-2 at 500 K. PCl5(g) PCl3(g) + Cl2(g) A reaction m...
Questions

Chemistry, 05.07.2020 19:01

Mathematics, 05.07.2020 19:01


Mathematics, 05.07.2020 20:01

Health, 05.07.2020 20:01

Mathematics, 05.07.2020 20:01

Advanced Placement (AP), 05.07.2020 20:01

Mathematics, 05.07.2020 20:01


Mathematics, 05.07.2020 20:01

Advanced Placement (AP), 05.07.2020 20:01

Advanced Placement (AP), 05.07.2020 20:01

Mathematics, 05.07.2020 20:01

Social Studies, 05.07.2020 20:01

Mathematics, 05.07.2020 20:01

Mathematics, 05.07.2020 20:01

Mathematics, 05.07.2020 20:01


Mathematics, 05.07.2020 20:01

![Qc=\frac{[C]^{c} *[D]^{d} }{[A]^{a} *[B]^{b} }](/tpl/images/0689/6440/ada98.png)
![Qc=\frac{[PCl_{3}]*[Cl_{2} ]}{[PCl_{5} ] }](/tpl/images/0689/6440/81d8b.png)
![[PCl_{3} ]=\frac{2.60*10^{-2} moles}{1 liter} =2.60*10^{-2} \frac{moles}{L}](/tpl/images/0689/6440/fd446.png)
![[Cl_{2} ]=\frac{3.82*10^{-2} moles}{1 liter} =3.82*10^{-2} \frac{moles}{L}](/tpl/images/0689/6440/fced6.png)
![[PCl_{5} ]=\frac{0.121 moles}{1 liter} =0.121 \frac{moles}{L}](/tpl/images/0689/6440/529d8.png)