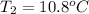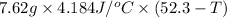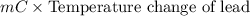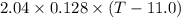Chemistry, 19.06.2020 00:57 straightbarz5916
A 2.04 g lead weight, initially at 10.8 oC, is submerged in 7.62 g of water at 52.3 oC in an insulated container. clear = 0.128 J/g oF; water = 4.18 J/goC. What is the final temperature of both the weight and the water at thermal equilibrium

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Iknow the answer to 13 is b and 14 is d. i just need to know why the correct answers are correct
Answers: 3


Chemistry, 23.06.2019 10:00
How many grams of cupric sulfate pentahydrate are needed to prepare 50.00 ml of 0.0800m cuso4× 5h2o?
Answers: 3

Chemistry, 23.06.2019 17:00
Anonaqueous solution has a solvent that is not water. which is an example of a nonaqueous solution?
Answers: 2
You know the right answer?
A 2.04 g lead weight, initially at 10.8 oC, is submerged in 7.62 g of water at 52.3 oC in an insulat...
Questions



Mathematics, 25.06.2021 02:30






Business, 25.06.2021 02:30


Physics, 25.06.2021 02:30




Mathematics, 25.06.2021 02:30

Chemistry, 25.06.2021 02:30




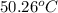 .
.