
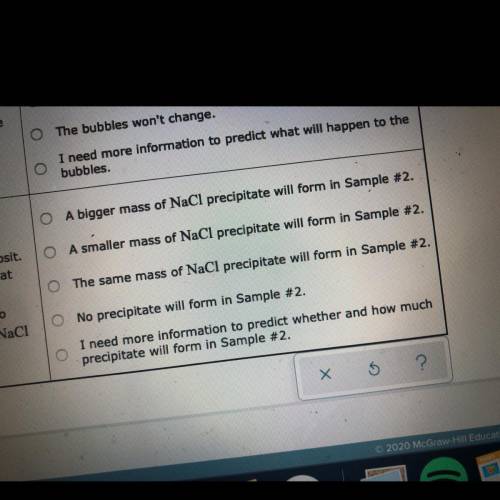
Two 250 mL samples of water are drawn from a deep
well bored into a large underground salt (NaCl) deposit. Sample #1 is from the top of the well, and is initially at 42 °C. Sample #2 is from a depth of 150 m, and is
initially at 8 °C. Both samples are allowed to come to room temperature (20 °C) and 1 atm pressure. An NaCl precipitate is seen to form in Sample #1.
The rest of the question is in the picture!

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:30
The climate of the continental united states is generally 1. tropical 2. temperate 3. arctic 4. highland
Answers: 1

Chemistry, 22.06.2019 22:00
The diagrams to the right show the distribution and arrangement of gas particles in two different containers. according to kinetic-molecular theory, which of the following statements is true? check all that apply. if the temperatures of both containers are equal, container a has greater pressure than container b. if the volume of container a decreased, its pressure would decrease. if the pressure in both containers is equal, container a has a lower temperature than container b.
Answers: 2

Chemistry, 23.06.2019 01:30
If a particle has z = 25 and 23 electrons, what is its charge?
Answers: 2

Chemistry, 23.06.2019 03:50
How many liters of oxygen gas, at standardtemperature and pressure, will react with 35.8 grams ofiron metal? 4 fe (s) + 3 o2 (g) → 2 fe2o3 (s)
Answers: 3
You know the right answer?
Two 250 mL samples of water are drawn from a deep
well bored into a large underground salt (NaCl) d...
Questions

Advanced Placement (AP), 02.10.2019 21:30

History, 02.10.2019 21:30


Social Studies, 02.10.2019 21:30

Mathematics, 02.10.2019 21:30


Chemistry, 02.10.2019 21:30

Social Studies, 02.10.2019 21:30


Mathematics, 02.10.2019 21:30

Mathematics, 02.10.2019 21:30




Health, 02.10.2019 21:30

English, 02.10.2019 21:30



Spanish, 02.10.2019 21:30

Social Studies, 02.10.2019 21:30



