
Chemistry, 18.06.2020 06:57 ynclankaedon
What volume of 0.300 M Cu(OH)2 solution is needed to react with 500. mL of 0.100 M H3PO4 solution?
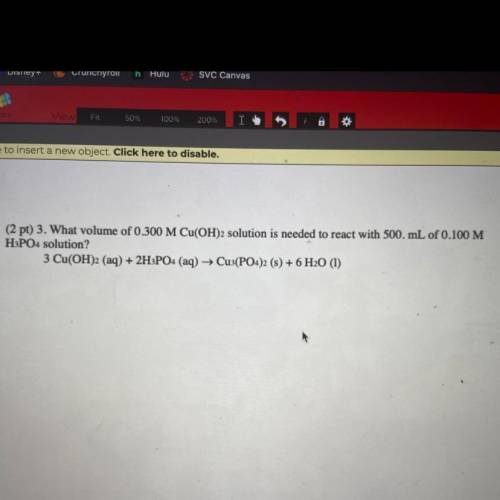

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
This chart represents the melting point of several substance. what besy explains the high melting point of the salt?
Answers: 2

Chemistry, 22.06.2019 14:30
Is a pencil falling to the floor anon contact force, a force, or a contact force
Answers: 1

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3
You know the right answer?
What volume of 0.300 M Cu(OH)2 solution is needed to react with 500. mL of 0.100 M H3PO4 solution?
<...
Questions


Mathematics, 18.07.2019 17:00

English, 18.07.2019 17:00

Physics, 18.07.2019 17:00



Mathematics, 18.07.2019 17:00



Biology, 18.07.2019 17:00

Mathematics, 18.07.2019 17:00

Mathematics, 18.07.2019 17:00


Chemistry, 18.07.2019 17:00

History, 18.07.2019 17:00

Biology, 18.07.2019 17:00


History, 18.07.2019 17:00

History, 18.07.2019 17:00



