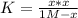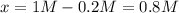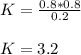Chemistry, 18.06.2020 05:57 andregijoe41
PCl5 dissociates according to the reaction: PCl5(g) = PCl3(g) + Cl2(g) One mole of PCl5 was placed in one liter of solution. When equilibrium was established, 0.2 mole of PCl5 remained in the mixture. What is the equilibrium constant for this reaction? (Hint: remember the ICE procedure? initial, change, and equilibrium)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:30
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1

Chemistry, 22.06.2019 08:30
Sally is making a model of a magnesium atom with an atomic mass number of 24 for her chemistry class. she has foam balls for the protons, neutrons, and electrons. she has added 6 neutrons to her model so far. how many more neutrons does she need to add to complete her neutral atom of magnesium?
Answers: 1


Chemistry, 22.06.2019 17:00
Complete each row of the table below by filling in the missing prefix or missing exponent.
Answers: 1
You know the right answer?
PCl5 dissociates according to the reaction: PCl5(g) = PCl3(g) + Cl2(g) One mole of PCl5 was placed i...
Questions


English, 07.09.2020 03:01


Mathematics, 07.09.2020 03:01


History, 07.09.2020 03:01




Mathematics, 07.09.2020 03:01

Mathematics, 07.09.2020 03:01



World Languages, 07.09.2020 03:01





Mathematics, 07.09.2020 03:01

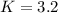
![K=\frac{[PCl_3][Cl_2]}{[PCl_5]}](/tpl/images/0688/6864/bc353.png)
 due to the reaction extent (ICE procedure):
due to the reaction extent (ICE procedure):