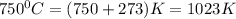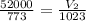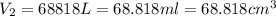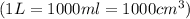Chemistry, 18.06.2020 03:57 jadajones5313
A hot air balloonist puts 52000 L of air into their balloon at 500 Celsius and 975 atm. When they heat the air to 750 celsius what is the final volume (in cm^3) in the balloon?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3

Chemistry, 23.06.2019 06:00
Complete the sentences to best explain the ranking.match the words below to the appropriate blanks in the sentences.a less polar bondhigher molar massion-dipole forcesstronger intermolecular forcesdipole-dipole forcesdispersion forceshydrogen bonding1. h2s and h2se exhibit the following intermolecular forces:.2. therefore, when comparing h2s and h2se the one with a has a higher boiling point .3. the strongest intermolecular force exhibited by h2o is . therefore, when comparing h2se and h2o the one with has a higher boiling point.
Answers: 1

Chemistry, 23.06.2019 08:40
Calculate the number of grams of sodium in 3.00 g of each sodium-containing food additive.
Answers: 3

Chemistry, 23.06.2019 10:30
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3 2 kilopascals, what is the partial pressure of the hydrogen?
Answers: 1
You know the right answer?
A hot air balloonist puts 52000 L of air into their balloon at 500 Celsius and 975 atm. When they he...
Questions

Biology, 10.11.2020 23:00


Mathematics, 10.11.2020 23:00

Mathematics, 10.11.2020 23:00

Mathematics, 10.11.2020 23:00

History, 10.11.2020 23:00


Business, 10.11.2020 23:00

Mathematics, 10.11.2020 23:00

Mathematics, 10.11.2020 23:00


History, 10.11.2020 23:00

Mathematics, 10.11.2020 23:00


English, 10.11.2020 23:00


English, 10.11.2020 23:00

Medicine, 10.11.2020 23:00

Chemistry, 10.11.2020 23:00

Computers and Technology, 10.11.2020 23:00

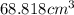
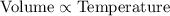
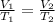 (At constant pressure and number of moles)
(At constant pressure and number of moles)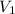 = initial volume = 52000 L
= initial volume = 52000 L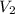 = final volume = ?
= final volume = ?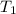 = initial temperature =
= initial temperature = 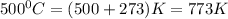
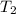 = final temperature =
= final temperature =